Contact Dr. Lu for information about cancer treatments。聯繫盧博士,獲取有關癌症治療資訊。
Potential harm from Tropicamide eye drops | 托吡卡胺滴眼液的潛在危害
Contact Dr. Lu for information about cancer treatments。聯繫盧博士,獲取有關癌症治療資訊。
When you go to an eye doctor (optometrist) for a vision examination, he might use tropicamide eye drops without consulting you for your permission. The practice that doctors do whatever without your consent is common, which is good for their business but not necessarily good for your health.
Recently, someone went for a vision exam but did not mean to do any ophthalmological examination which should be done by an ophthalmologist. The optometrist wanted to conduct an ophthalmological examination for which Tropicamide eye drops was used. Like many other patients, he is reluctant to confront this optometrist. But the side effect is apparent. After use of this eye drops, the vision gets worse (blurred vision). He used to have very good near-sighted vision as an older man, and he can read books, text on phones and computer screens just fine. But the blurriness make the reading a bad experience.
Remember that all medications are toxic!
當你去看眼科醫生(驗光師)進行視力檢查時,他可能會在沒有徵得你同意的情況下使用托吡卡胺滴眼液。 醫生未經您同意就為所欲為的做法很普遍,這對他們的業務有利,但不一定對您的健康有益。
最近,有人去做視力檢查,但沒有做任何應該由眼科醫生做的眼科檢查的意思。 驗光師想要進行眼科檢查,為此使用了托吡卡胺滴眼液。 和許多其他患者一樣,他不願意與這位驗光師對質。 但副作用是顯而易見的。 使用這種眼藥水後,視力會變差(視力模糊)。 作為一個老人,他曾經有很好的近視視力,他可以很好地閱讀書籍、手機和電腦屏幕上的文字。 但是模糊使閱讀體驗很差。
請記住,所有藥物都是有毒的!
Jump to navigation Jump to search
 |
|
| Clinical data | |
|---|---|
| Trade names | Mydriacyl, others |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Pregnancy category |
|
| Routes of administration |
Topical eye drops |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 45% |
| Identifiers | |
| CAS Number | |
|---|---|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.014.673 |
| Chemical and physical data | |
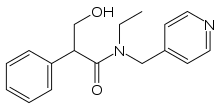
| Formula | C17H20N2O2 |
| Molar mass | 284.359 g·mol−1 |
| 3D model (JSmol) | |
| |
Tropicamide, sold under the brand name Mydriacyl among others, is a medication used to dilate the pupil and help with examination of the eye.[3] Specifically it is used to help examine the back of the eye.[4] It is applied as eye drops.[3] Effects occur within 40 minutes and last for up to a day.[3]
Common side effects include blurry vision, increased intraocular pressure, and sensitivity to light.[3] Another rare but severe side effect is psychosis, particularly in children.[3] It is unclear if use during pregnancy is safe for the fetus.[5] Tropicamide is in the antimuscarinic part of the anticholinergic family of medications.[3] It works by making the muscles within the eye unable to respond to nerve signals.[3]
Tropicamide was approved for medical use in the United States in 1960.[3] It is on the World Health Organization’s List of Essential Medicines.[6]
Medical use
Tropicamide is an antimuscarinic drug that produces short acting mydriasis (dilation of the pupil) and cycloplegia[7] when applied as eye drops. It is used to allow better examination of the lens, vitreous humor, and retina. Due to its relatively short duration of effect (4–8 hours), it is typically used during eye examinations such as the dilated fundus examination, but it may also be used before or after eye surgery. Cycloplegic drops are often also used to treat anterior uveitis, decreasing risk of posterior synechiae and decreasing inflammation in the anterior chamber of the eye.
Tropicamide is occasionally administered in combination with p-hydroxyamphetamine (brand name Paremyd), which is a sympathomimetic. The use of the sympathomimetic drug causes the iris dilator muscle to be directly stimulated, causing increased dilation. In the United States, the sympathomimetic drop most commonly used along with tropicamide, is 2.5% phenylephrine hydrochloride (brand name AK-Dilate).
Side effects
Tropicamide induces transient stinging and a slight and transient rise in intraocular pressure in the majority of patients. It may cause redness or conjunctivitis (inflammation) and also blurs near vision for a short while after instillation (care must be taken, and the patient must only drive when vision returns to normal). Tropicamide may, in very rare cases,[8] cause an attack of acute angle-closure glaucoma. This tends to be in patients with narrow anterior chamber angles, and closure risk must be assessed by the practitioner prior to instillation.
Tropicamide is often preferred to atropine because atropine has a longer half-life, causing prolonged dilation and blurry vision for up to a week. Atropine has less sting effect, but can be toxic or fatal if ingested in large quantities by children or adults.
With eye drops, systemic effects are minimal to nonexistent due to very low absorption into the bloodstream.[9]
Illicit use
According to the researchers of the European Commission-funded ReDNet Project, in Russia tropicamide is currently abused (injected intravenously) as an inexpensive recreational deliriant drug.[10][11]
Stereochemistry
Tropicamide has a chiral center and two enantiomers. Medications are racemates.[12]
| Enantiomers | |
|---|---|
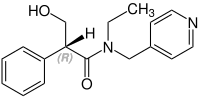 (R)-Tropicamid CAS: 92934-63-9 |
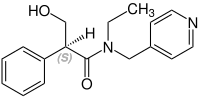 (S)-Tropicamid CAS: 92934-64-0 |
References
- “Summary for ARTG Entry: 25356 Mydriacyl tropicamide 0.5% eye drops bottle”. Retrieved 29 July 2020.[permanent dead link]
- “Mydriacyl 1% eye drops, solution – Summary of Product Characteristics (SmPC)”. (emc). 12 February 2020. Retrieved 29 July 2020.
- “Tropicamide”. The American Society of Health-System Pharmacists. Archived from the original on 28 December 2016. Retrieved 8 December 2016.
- World Health Organization (2009). Stuart MC, Kouimtzi M, Hill SR (eds.). WHO Model Formulary 2008. World Health Organization. p. 314. hdl:10665/44053. ISBN 9789241547659.
- “Tropicamide ophthalmic Use During Pregnancy”. Drugs.com. Archived from the original on 28 December 2016. Retrieved 28 December 2016.
- World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- Manny RE, Hussein M, Scheiman M, Kurtz D, Niemann K, Zinzer K (July 2001). “Tropicamide (1%): an effective cycloplegic agent for myopic children”. Investigative Ophthalmology & Visual Science. 42 (8): 1728–35. PMID 11431435. Archived from the original on 2013-01-12.
- Liew, Gerald; Mitchell, Paul; Wang, Jie Jin; Wong, Tien Yin (January 2006). “Fundoscopy: to dilate or not to dilate?”. BMJ (Clinical Research Ed.). 332 (7532): 3. doi:10.1136/bmj.332.7532.3. PMC 1325111. PMID 16399709.
- Vuori, Marja-Liisa; Kaila, Timo; Iisalo, Esko; Saari, K. Matti (1994-01-01). “Systemic Absorption and Anticholinergic Activity of Topically Applied Tropicamide”. Journal of Ocular Pharmacology and Therapeutics. 10 (2): 431–437. doi:10.1089/jop.1994.10.431. ISSN 1080-7683. PMID 8083562.
- Bersani FS, Corazza O, Simonato P, Mylokosta A, Levari E, Lovaste R, Schifano F (2013). “Drops of madness? Recreational misuse of tropicamide collyrium; early warning alerts from Russia and Italy”. General Hospital Psychiatry. 35 (5): 571–3. doi:10.1016/j.genhosppsych.2013.04.013. PMID 23706777.
- Krokodil: The drug that eats junkies Archived 2017-09-22 at the Wayback Machine
Rote Liste (in German). Vol. 57. Frankfurt/Main: Rote Liste Service GmbH. 2017. p. 224. ISBN 978-3-946057-10-9. Arzneimittelverzeichnis für Deutschland (einschließlich EU-Zulassungen und bestimmter Medizinprodukte)

